 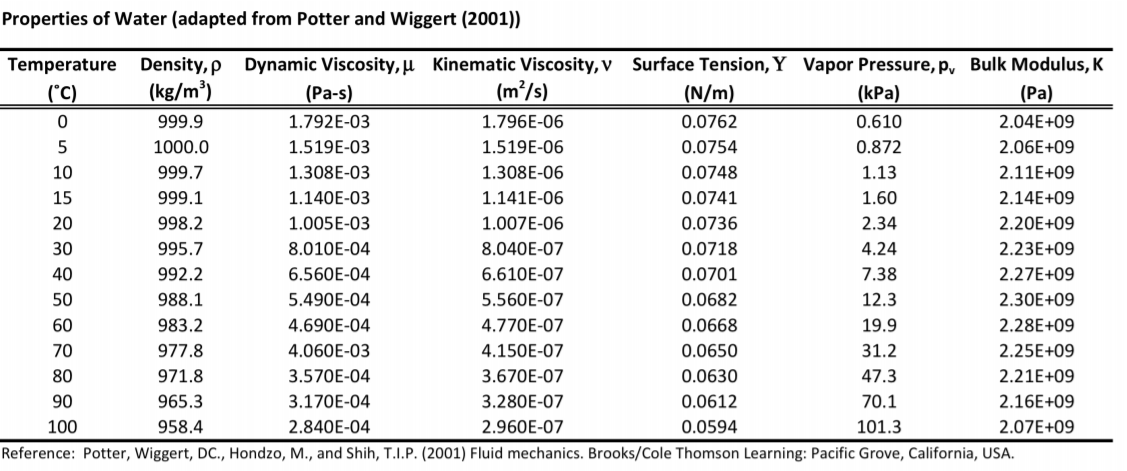 
25 Water is a tasteless, odorless liquid at ambient temperature and pressure. The uncertainty estimation of one of the reports included in the analysis has been re-evaluated. Physical properties Water is the chemical substance with chemical formula H 2O one molecule of water has two hydrogen atoms covalently bonded to a single oxygen atom. The STP and the SATP should not be confused with the standard state commonly used in thermodynamic evaluations of the Gibbs energy of a reaction. This paper determines the formulae of density and relative density, with their uncertainties, in the temperature range 0 ☌ to 40 ☌. An unofficial, but commonly used standard is standard ambient temperature and pressure (SATP) as a temperature of 298.15 K (25 C, 77 F) and an absolute pressure of 100 kPa (14.504 psi, 0.987 atm). In chemistry, IUPAC established standard temperature and pressure (informally abbreviated as STP) as a temperature of 273.15 K (0 C, 32 F) and an absolute pressure of 100 kPa (14.504 psi, 0.987 atm, 1 bar). The International Standard Metric Conditions for natural gas and similar fluids are 288.15 K (59.00 F 15.00 C) and 101.325 kPa. NIST uses a temperature of 20 C (293.15 K, 68 F) and an absolute pressure of 101.325 kPa (14.696 psi, 1 atm). Compute the Depth of Water associated with a Pressure The similarities in mechanical behavior between M666-D and M666-W suggest that any effect of water on dislocation glide is inconsequential at room temperature.The Density of Water at Standard Temperature and Pressure (STP) is 998.2071 kg/m 3 in SI units. For example, at 0☌ the density of water is 0.9998 g/mL, but at 80☌ the density is 0. Water density increases as the temperature gets colder. The density of pure water is altered by temperature. The specific volume of water depends on temperature as shown below: Heavy. Although the density of water is pretty close to 1 g/mL, certain disciplines of science need to know the density of water with a higher specificity. Usually the solid form of a pure substance is denser than the liquid form of the same substance. Density is an intensive physical property, meaning that it does not change with sample size. Pure substances have a unique density at a given temperature. The output specific volume is given as cm /g, ft /lb, gal (US liq)/lb and ft. Density can be used to identify a substance. This is used as a reference substance for defining Specific Gravity. The calculator below can be used to calculate the liquid water specific volume at given temperatures. While calculating the molecular mass of water it is the sum of the atomic mass of all the elements present in the molecule.The Density of Water at Standard Temperature and Pressure is 62.316 lbs/ft 3. Note: The exact density of water is not 1 g/ml, but a bit less of 0.099983 g/ml at $4^0$Celsius. There are two hydrogen atoms and one atom of oxygen in each water molecule as depicted by its molecular formula $$. We need to know the chemical formula of water to calculate the number of molecules and atoms in a water drop. This question evolves understanding the concept of mole and molar volume which is why we know that molar volume of all gases at STP is 22.4 L. Now since one mole molecules of all gases contain the same number of molecules therefore they occupy the same molecules under STP. Hint: We know that avagadro’s law explains that equal volumes of all gases contain equal numbers of molecules under similar conditions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
